
1 / 3






| Still deciding? Get samples of $ ! US$ 1/Piece Request Sample |

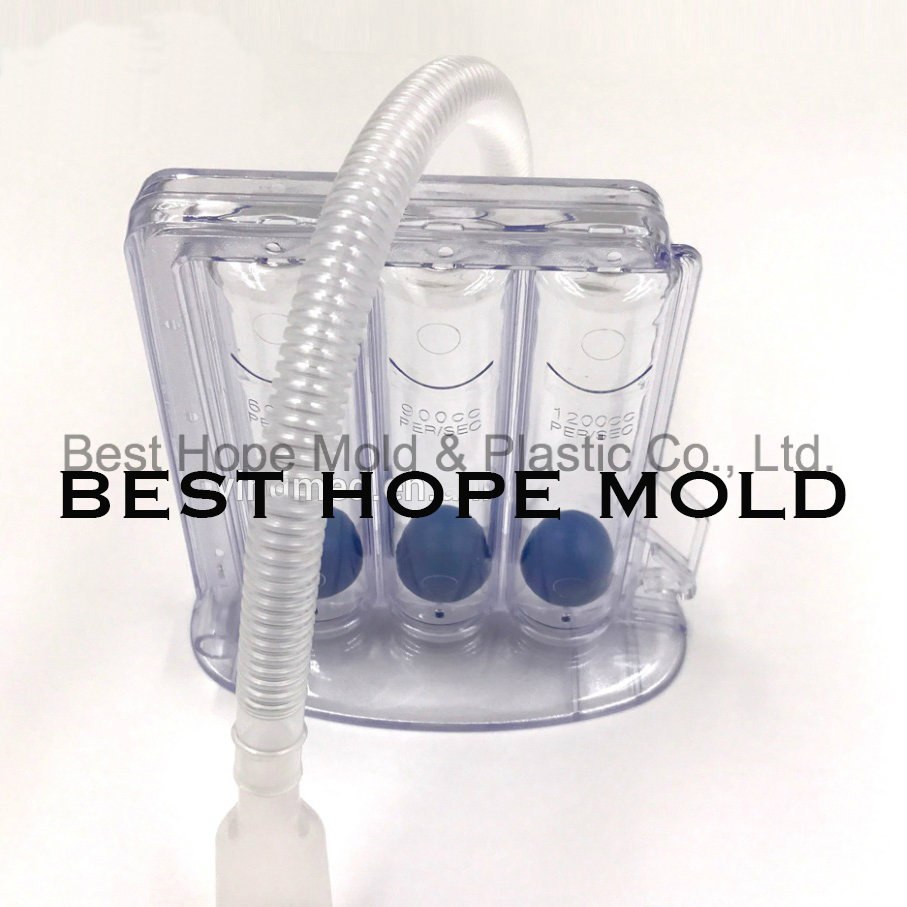
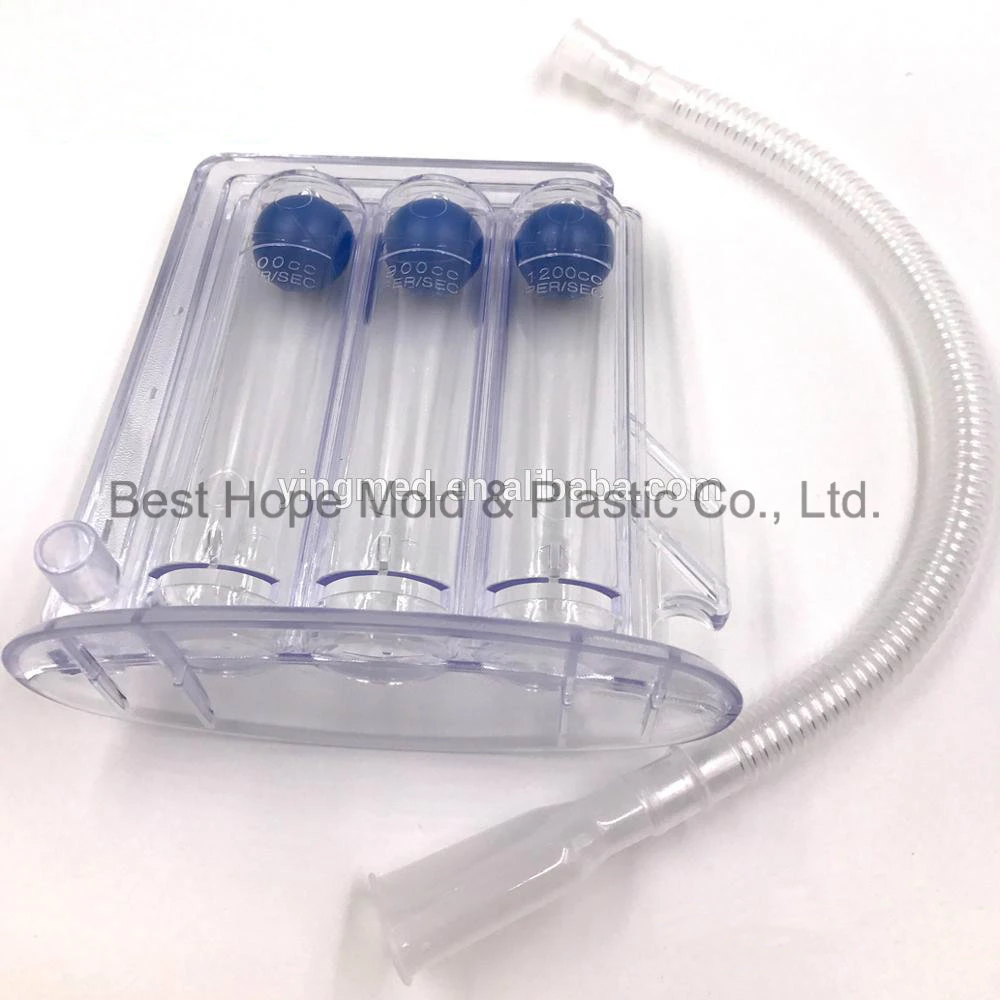
| Description | Plastic injection mold for Mould for 3 Balls Incentive Spirometer |
| Part Material | ABS, PC, PP, PE, PA, TPE, POM, PBT, PEEK, PPO, PEI, PES etc. |
| Cavity Number | 1/2/4/8/16/32/48/64...(optional) |
| Runner | Hot runner or cold runner |
| Core & Cavity Material | S136, 1.2083 stainless steel |
| Mould Standard | MISUMI, DME, CUMSA, HASCO |
| Mould Life | Long mold life |
| Available Format | .stp/.pdf/.x-t/.step./.dwg/.dxf/.igs/.prt/.stl/.sldprt |


We are an ISO13485 certified manufacturing factory specializing in precision mold design, manufacturing, and high-end plastic injection molding. We focus on precision tooling for Medical Devices with rich experience in medical products.


Medical moulds from regular medical disposables to high precise minimally invasive devices, Communication Parts, Household Appliances Parts, Electronics Parts, Cosmetics Parts etc.



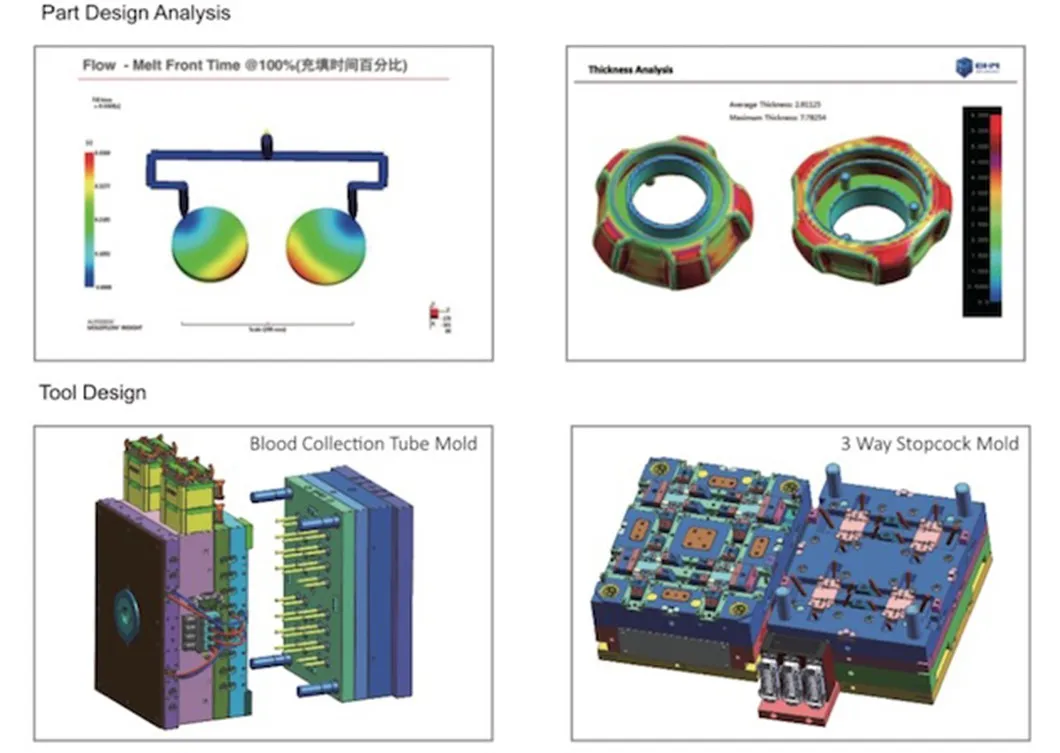

Before shipment, we perform rigorous tests, inspections, and take photos. All spare parts and drawings are packed in a secure wood box.
We regularly attend international medical exhibitions such as CMEF, MEDICA, Arab Health, and FIME.


 Emy Car
Emy Car